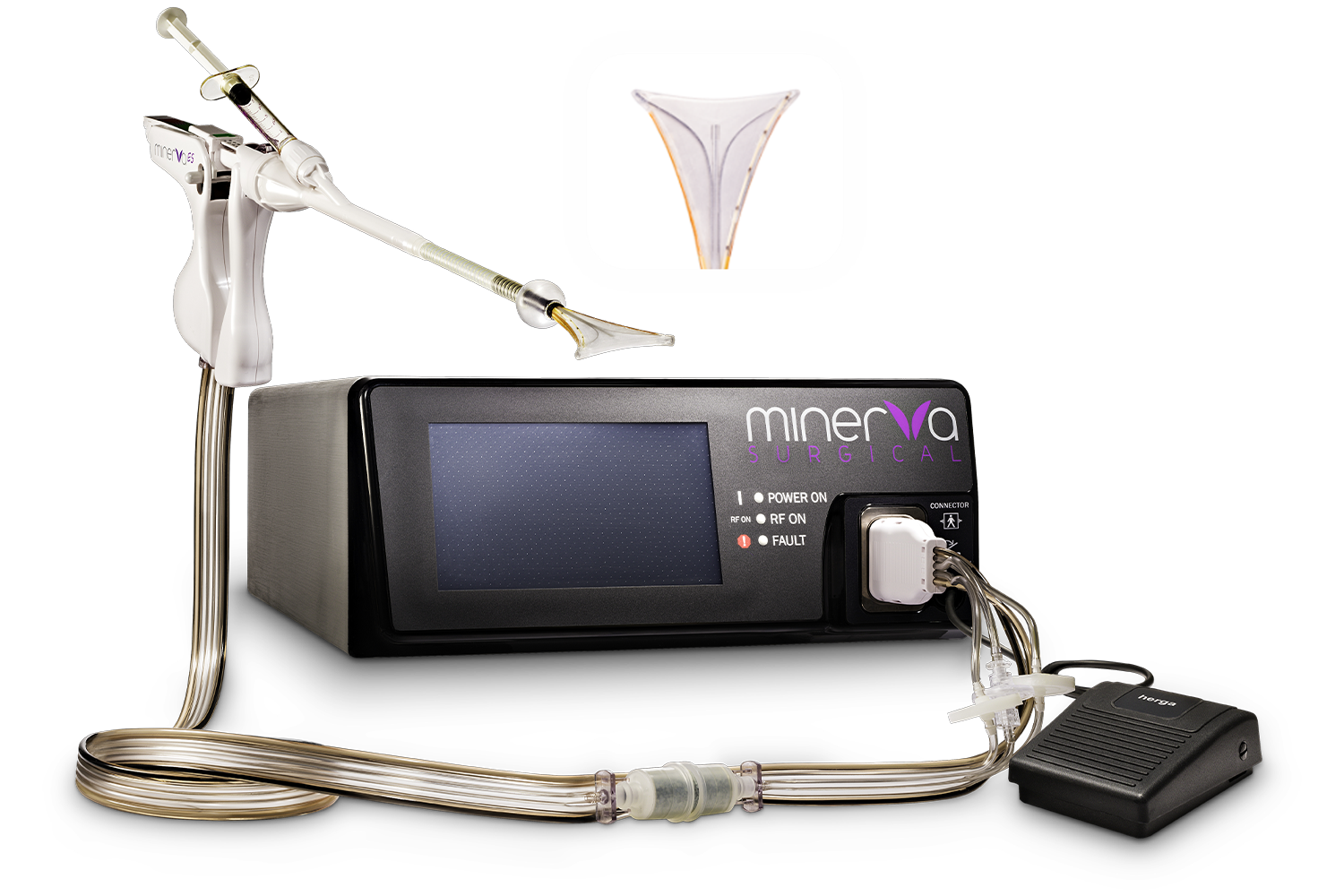

The Minerva Endometrial Ablation System (EAS) is engineered with a unique, triple-mechanism approach to endometrial ablation. By utilizing three distinct forms of energy (simultaneous use of bipolar RF energy, direct-contact thermal tissue ablation, and the use of heated intracavitary fluids), the system ensures a comprehensive and uniform treatment of the uterine cavity.
During the ablation cycle, plasma seeks out the least ablated tissue or tissue that offers the lowest resistance–or does not impede–plasma energy. For complete coverage, heated intrauterine cavity fluids flow throughout the sealed cavity, ablating tissue not in direct contact with the array.
As tissue is desiccated, resistance–or impedance–increases in that area. In response, the plasma changes course and seeks the least ablated tissue or tissue that offers the lowest resistance.
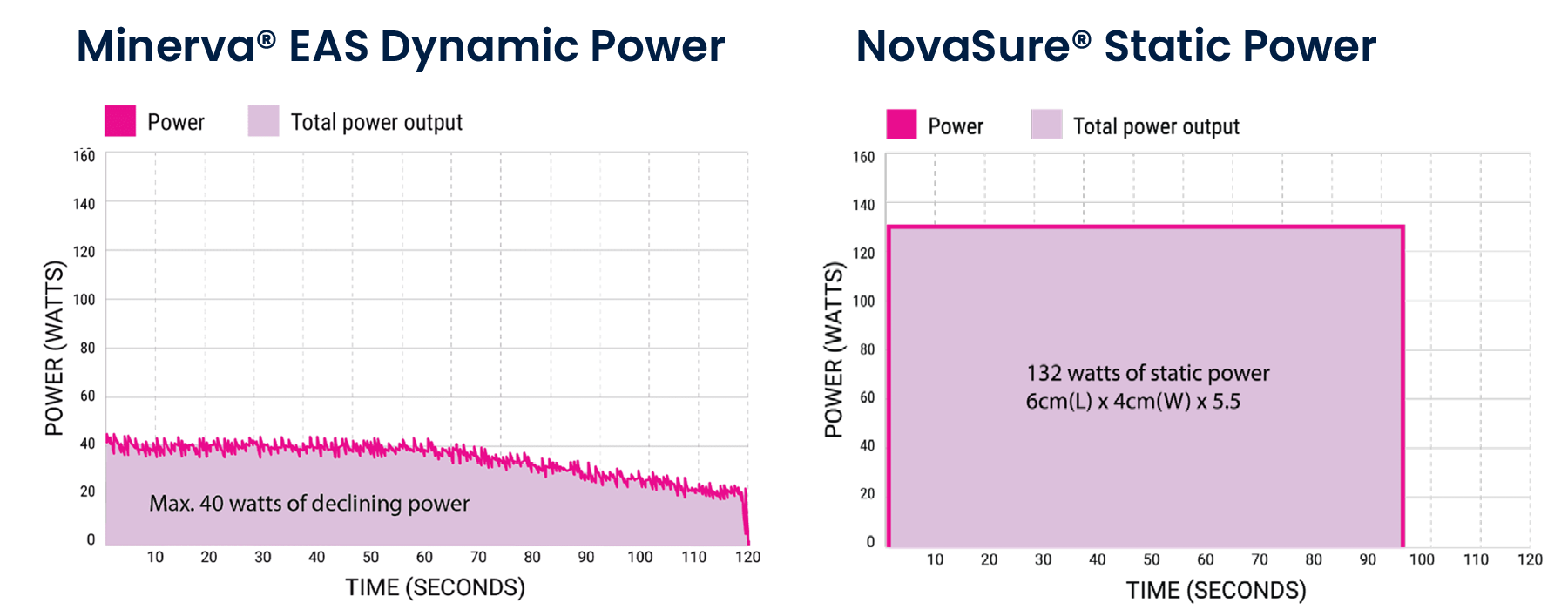
Our advanced controller continuously monitors the uterine cavity (up to 50 times per second) to dynamically direct plasma to the least ablated tissue. In response, the power output automatically decreases in real-time as the tissue impedance increases.
Starting with just 40 watts of declining power prevents the uterine cavity from being overwhelmed with energy, which may result in an early impedance shut off with a classic RF ablation.
In 120 seconds, a complete and consistent depth of ablation is achieved.
The videos below show actual Minerva EAS ablation data as collected in real time by a graphical data software program for comprehensive analysis of data. With the assistance of an Axora Medical Sales Representative, this data can be available to your team during treatments.
The red line shows the power dynamically responding to an increase in ablated surface area impedance by decreasing accordingly. The yellow line represents increasing impedance, and together they show the power output declining as the area of ablated tissue increases. The cavity receives the minimum amount of power required to ablate from start to finish–far less than the classic RF ablation.



Results from different clinical investigations are not directly comparable.
Information provided for educational purposes only.
1. NovaSure V5 Device Instructions for Use, Revision 003. Hologic, https://www.hologic.com/package-inserts/gyn-surgical-products/novasure-endometrial-ablation?_gl=1*fjxi2n*_gcl_au*OTk2MDE5OTE2LjE3MzgxODgwODA. Accessed 22 Apr. 2025.
2. Minerva Single Arm Clinical Study, Minerva Endometrial Ablation System Instructions for Use, L0107 Rev. F. Axora Medical.
For your convenience we’ve selected a series of our most viewed videos. Please visit the Resource Center for access to our complete video library.

Every patient will receive a 120 second treatment cycle, however, the amount of energy delivered during the 120 seconds will vary from patient to patient depending on uterine cavity size and thickness of the endometrium. Each ablation starts with a maximum of 40W of power and declines over the course of the ablation as the impedance within the cavity increases.

The Minerva EAS does not rely solely on direct tissue contact to achieve optimal ablation. Three simultaneous and complementary methods of tissue ablation are being employed by the Minerva EAS, providing a truly global ablation.

The Minerva EAS is the only ablation system that modulates the power level over the course of the ablation cycle. The controller adjusts power in real time by assessing the changes in the state of the uterine cavity during the ablation process, with measurements being taken and adjustments made 50 times per second. During the ablation process, the area of unablated tissue decreases, and power starts dropping accordingly, with plasma being redirected.
Thus focusing ONLY on the least ablated endometrium for a uniform surface area coverage and depth of ablation.

Plasma is another term for ionized gas, in this case argon. The argon gas inside the membrane is ionized by the RF energy delivered to the handpiece from the Minerva Controller.

The cervix should be dilated. An 8mm Hegar dilator is included in the device packaging. The dilator has graduated length markings to ensure accurate measurement of the cervix.

Do not turn off the Controller. Most error codes may be resolved via troubleshooting or replacing the handpiece. Then the Controller will proceed through the safety checks and resume and ablate for the remaining treatment time. In rare instances, a hard fault may be encountered that would require cycling off power to the Controller. Consult the Operator’s Manual or call Axora Medical at 855-646-7874 for more information.

The cervical sealing balloon can be inflated a maximum of 2 times. If a cervical seal still cannot be achieved, reassess to make sure the device is properly positioned in the uterine cavity and rule out uterine perforation.

In a clinical study, patients who underwent treatment with the Minerva EAS achieved an amenorrhea rate of 72%, or 7 out of 10 women, at 1-year followup1.
1. Laberge P, Garza-Leal J, Fortin C, Grainger D, Johns DA, Adkins RT, Presthus J, Basinski C, Swarup M, Gimpelson R, Leyland N, Thiel J, Harris M, Burnett PE, Ray GF. A Randomized Controlled Multicenter US Food and Drug Administration Trial of the Safety and Efficacy of the Minerva Endometrial Ablation System: One-Year Follow-Up Results. J Minim Invasive Gynecol. 2017 Jan 1;24(1):124-132. doi: 10.1016/j.jmig.2016.09.009. Epub 2016 Sep 26. PMID: 27687851.

At three years following the procedure, the Minerva EAS treated patients had a 0.9% hysterectomy rate.2 While studies from different clinical investigations are not comparable, for educational purposes only, in their own separate clinical study NovaSure reported a 6.3% hysterectomy rate at 3 years in its IFU.3
2. Data on file, Minerva Single Arm Clinical Study, Minerva Endometrial Ablation System Instructions for Use, L0107 Rev. F. Axora Medical.
3. NovaSure V5 Device Instructions for Use, Revision 003. Hologic, https://www.hologic.com/package-inserts/gyn-surgical-products/novasure-endometrial-ablation?_gl=1*fjxi2n*_gcl_au*OTk2MDE5OTE2LjE3MzgxODgwODA. Accessed 17 Mar. 2026.

The argon Canister will last for approximately 7 treatments. A CO2 Canister will last for approximately 25 treatments.

No. According to the product labeling, use of the Minerva EAS, and all other second-generation endometrial ablation technologies, is contraindicated in a patient with any anatomic condition that could lead to weakening of the myometrium. Please consult instructions for use for complete indications, contraindications, warnings and precautions.

The Minerva treatment should not be performed concomitantly with the placement of the Essure device. The safety and effectiveness of the Minerva treatment has not been evaluated in patients with the Essure device.
With a comprehensive portfolio of minimally invasive solutions spanning diagnosis through treatment, Axora Medical gives you more options to support personalized patient care.
Schedule a demoWith a comprehensive portfolio of minimally invasive solutions spanning diagnosis through treatment, Axora Medical gives you more options to support personalized patient care.
Schedule a demo